We explain what the boiling point is and how it is calculated. Examples of boiling point. Melting and freezing point.

What is boiling point?
He boiling point It is the temperature at which the vapor pressure of the liquid (pressure exerted by the gas phase on the liquid phase in a closed system at a certain temperature) is equal to the pressure surrounding the liquid. When this occurs, the liquid transforms into a gas.
The boiling point is a property that depends strongly on environmental pressure . A liquid subjected to a very high pressure will have a higher boiling point than if we subject it to lower pressures, that is, it will take longer to turn into vapor when it is subjected to high pressures. Due to these variations in the boiling point, the IUPAC defined the standard boiling point: it is the temperature at which a liquid turns into a vapor at a pressure of 1 bar.
An important issue is that The boiling point of a substance cannot be increased indefinitely . When we increase the temperature of a liquid until it passes its boiling point and still continue to increase it, we reach a temperature called “critical temperature”. The critical temperature is the temperature above which the gas cannot be transformed into a liquid by increasing pressure, that is, it cannot be liquefied. At this temperature, there is no defined liquid phase or vapor phase.
The boiling point is different for each substance. This property depends on the molecular mass of the substance and the type of intermolecular forces it presents (hydrogen bonds, permanent dipole, induced dipole), which in turn depends on whether the substance is polar covalent or nonpolar covalent (nonpolar).
When the temperature of a substance is below its boiling point, only a portion of its molecules located on its surface will have enough energy to break the surface tension of the liquid and escape into the vapor phase. On the other hand, when heat is supplied to the system, there is an increase in the entropy of the system (tendency to disorder of the system's particles).
How is the boiling point calculated?
Through the Clausius-Clapeyron equation phase transitions of a system composed of a single component can be characterized. This equation can be used to calculate the boiling point of substances and is applied as follows:
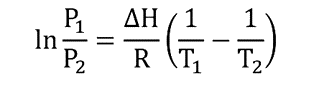
Where:
Q1 is the pressure equal to 1 bar, or in atmospheres (0.986923 atm)
T1 is the boiling temperature (boiling point) of the component, measured at the pressure of 1 bar (P1) and expressed in degrees Kelvin (K).
Q2 is the vapor pressure of the component expressed in bar or atm.
T2 is the temperature of the component (expressed in degrees Kelvin) at which the vapor pressure P is measured2.
𝚫H is the average vaporization enthalpy variation in the temperature range in which the calculation is being made. It is expressed in J/mol or equivalent energy units.
R is the gas constant equivalent to 8.314 J/Kmol
ln is the natural logarithm
The boiling temperature (boiling point) T is cleared1
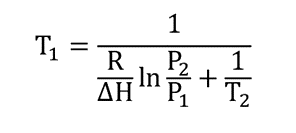
Examples of boiling point
Some recorded and known boiling points under normal pressure conditions (1 atm) are the following:
- Water: 100 ºC
- Helium: -268.9 ºC
- Hydrogen: -252.8 ºC
- Calcium: 1484 ºC
- Beryllium: 2471 ºC
- Silicon: 3265 ºC
- Carbon in the form of graphite: 4827 ºC
- Boron: 3927 ºC
- Molybdenum: 4639 ºC
- Osmium: 5012 ºC
- Tungsten: 5930 ºC
Melting point

The temperature at which a solid transforms into a liquid It is called the melting point and during the solid-liquid phase transition the temperature remains constant. In this case, heat is supplied to the system until its temperature increases enough so that the movement of its particles in the solid structure is greater, which causes them to separate and flow into the liquid phase.
The melting point also depends on pressure and is usually equal to the freezing point of matter (at which when sufficiently cooled a liquid becomes a solid) for most substances.
Continue in: Melting point
Freezing point
The freezing point is the opposite of the melting point, that is, the temperature at which a liquid contracts its particles lose movement and acquire a more rigid structure, resistant to deformation and with shape memory (unique of substances in a solid state). That is, it is the temperature at which the liquid transforms into a solid. Fusion requires supplying heat energy to the system, while freezing requires removing heat energy (cooling).
On the other hand, the freezing point also depends on the pressure . An example is what happens when water is cooled to a temperature of 0 ºC to 1 atm, when it freezes and becomes ice. If it is cooled to a pressure very different from 1 atm, the result could be very different, for example, if the pressure is much higher, it could take a while to freeze, as its freezing point decreases.
Melting and boiling point of water
Water is often used as a standard when measuring the melting and boiling points of substances. In general terms, At normal pressure, its boiling point is 100 ºC and its melting point is 0 ºC (in the case of ice). This can vary greatly in cases where the water has other substances dissolved in it, liquid or solid, as occurs with sea water, rich in salts, which modifies its physical and chemical properties.
The impact of pressure is also very noticeable. It is known that at 1 atm the boiling point of water is 100 ºC but taking it to 0.06 atm we would be surprised to notice that boiling occurs at 0 ºC (instead of freezing).
References
- Pedagogical manual of general chemistry practices on a microscale. Nemer, Beatriz Virginia Cervantes. Iberoamerican University (2006). ISBN 978-968-859-594-7.
- «Boiling Point» https://es.wikipedia.org/





